Despite excellent outcomes for Acute Promyelocytic Leukemia (APL) patients with combined ATRA and Arsenic Trioxide therapy, the underlying molecular mechanisms by which the PML::RARA fusion oncoprotein initiates APL are not yet clear. We recently found that the ability of PML::RARA to initiate aberrant serial replating (a surrogate for self-renewal) is dependent upon its ability to bind to DNA, since PML::RARA with a missense mutation (C88A) that prevents the binding of RARA to DNA eliminates aberrant replating. Currently, however, there is no consensus regarding the genomic binding sites of PML::RARA in myeloid progenitor cells and APL cells. Also, the transcriptional and epigenetic consequences of PML::RARA expression in primary hematopoietic cells remain unclear.
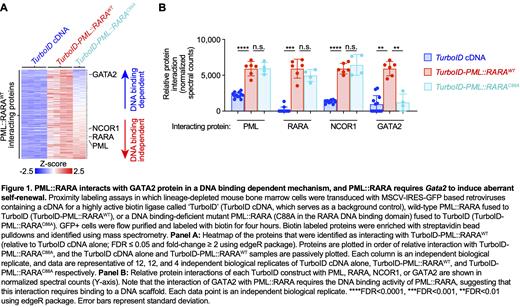
To define the genomic binding sites of PML::RARA, we generated V5 epitope tagged PML::RARA constructs in MSCV-based retroviral vectors, and transduced mouse hematopoietic stem progenitor cells (HSPCs) with retroviruses containing V5-PML::RARA WT, V5-PML::RARA C88A, or an empty vector. Anti-V5 ChIP-seq revealed that PML::RARA binds to 2,677 distinct loci in primary mouse hematopoietic cells, and 2,064 sites in primary human hematopoietic cells. We previously demonstrated that Gata2 expression is induced with PML::RARA, and indeed, we identified strong PML::RARA binding to the distal Gata2 enhancer in both human and mouse HSPCs. Surprisingly, ATAC-seq revealed that the vast majority of PML::RARA binding sites are found in regions that are chromatin-accessible even in WT promyelocytes, suggesting that these regions are “open” prior to PML::RARA expression, perhaps because of the normal binding of “pioneer” transcription factors to these regions in WT cells. Indeed, evaluation of transcription factor binding motifs in the regions occupied by PML::RARA revealed significant enrichment for RARA binding motifs, and also GATA motifs. We therefore performed ChIP-seq on GATA2 in HSPCs, which confirmed enrichment in binding at the same sites. These data suggested that PML::RARA and GATA2 may bind in close proximity at hundreds of loci in the genome. To test this, we performed proximity labeling studies followed by mass spectrometry using PML::RARA WT vs. PML::RARA C88A fused to the biotin ligase TurboID in primary HSPCs. This approach revealed that the interactions of several proteins with PML::RARA (including GATA2) are dependent on the binding of PML::RARA to DNA; this suggests that DNA may in fact act as a scaffold to stabilize these protein:protein interactions ( Figure 1).
To determine whether Gata2 may induce increased chromatin accessibility following PML::RARA expression, we performed ATAC-seq following Gata2 or PML::RARA overexpression in WT hematopoietic progenitor cells. 3,157 regions shared increased accessibility with either PML::RARA or Gata2 overexpression. Moreover, at a transcriptional level, we found that 535 (27.4%) of PML::RARA differentially expressed genes were coordinately regulated by Gata2 in WT cells. This suggests that PML::RARA indirectly regulates many of its gene targets via the upregulation of Gata2, and that Gata2 may be required for PML::RARA to initiate its transcriptional program. To test this, we performed serial replating assays using cells where Gata2 was knocked out either before or after PML::RARA expression. Consistent with our previous study, knock out of Gata2 following PML::RARA expression led to increased self-renewal capacity. However, knock out of Gata2 before PML::RARA expression prevented self-renewal. These data suggest a model in which GATA2 “opens” specific chromatin regions in early myeloid cells, which are then accessible to RARA or PML::RARA. The two proteins interact at these sites, tethered by their mutual interactions to their respective DNA binding motifs. PML::RARA binding promotes a pre-leukemic transcriptional program mediated in part by positively regulating Gata2. Gata2 is therefore initially required for PML::RARA to establish its transcriptional program. However, once that program is established, Gata2 paradoxically acts as a tumor suppressor, performing a normal role to limit proliferative excess in hematopoietic stem and progenitor cells.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal